甘油、糖、氨基酸合成肽催化剂 – LiTFSI(LiNTf2) 双三氟甲磺酰亚胺锂
This post is part of the series LiTFSI - 应用
Other posts in this series:
- 酯氨解反应催化剂 – LiTFSI(LiNTf2)双三氟甲烷磺酰亚胺锂
- 环氧开环反应催化剂 – LiNTf2(LiTFSI)双三氟甲烷磺酰亚胺锂
- 甘油、糖、氨基酸合成肽催化剂 – LiTFSI(LiNTf2) 双三氟甲磺酰亚胺锂 (Current)
关于LiNTf2产品可咨询默尼化工科技(上海)有限公司 www.ilschem.cn
View in Full Screen
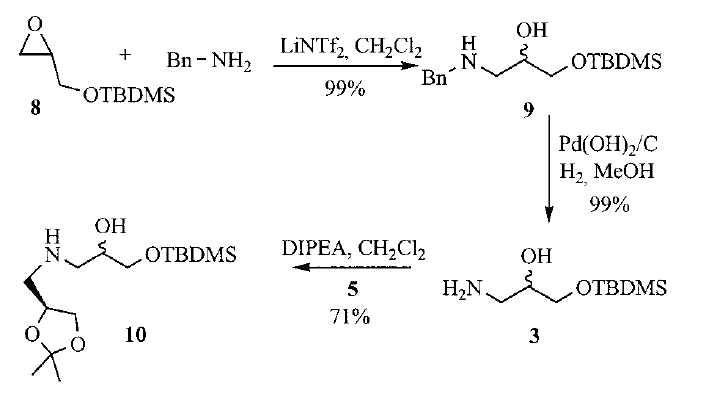
Synthesis of Glycidol- and Sugar-Derived Bicyclic β- and γ/δ-Amino Acids for Peptidomimetic Design
Constrained bicyclic β- and γ/δ-amino acids using glycidol and sugar derivatives were developed. The synthetic strategies involved epoxide ring opening of a glycidol derivative, and subsequent coupling with sugar-derived amines, leading to di- or trisubstitued bicyclic scaffolds after cyclisation with trifluoroacetic acid. Achievement of β- or γ/δ-amino acids was accomplished by changing the protecting group strategy of the starting materials. Compatibility of the scaffold with solid-phase peptide synthesis was assessed by preparing model peptidomimetics using acid- and base-labile resins, thus giving a new tool for peptidomimetic design.
Keywords: Scaffold / Peptidomimetics / Ring opening / Peptides / Amino acids / Solid-phase synthesis
Continue reading this series:
卓越的LiTFSI-三甘醇二甲醚全固态电池电解质
微信
支付宝